Unlike the other two sections, QA is a section that has a direct link to what you have done in school and college. Most of the topics that are tested on the CAT have also been a part of the school curriculum. This I feel is the biggest roadblock in front of test-takers wanting to achieve higher scores on the CAT Quant irrespective of their relationship with Quant, with high Math scores during X and XII exams not having any direct correlation with ability on the CAT QA.
This has to do with the simple fact that test-takers never fully grasp the difference between the two formats since they are as different from each other as chalk and cheese. So high is my resentment for the way they taught Math in school that I can write an entire blog post on that!
But I will try to condense my grouse to this — they never taught us to solve they taught us solutions; we never learned to solve, we memorized solutions.
If you want to get better at CAT Quant you should stop memorising solutions and start solving problems.
When I mean start solving problems I mean literally start solving a problem the way a mechanic will fix a bike.
– Do mechanics memorize the way they repaired each and every bike?
– Do mechanics start fixing a bike or car before they understand the problem?
– Do mechanics need to constantly revise the basics of how an automobile works before they begin to fix every new vehicle?
The answer to all the above questions is a resounding NO! So the first step is to make this perceptual shift in your mind before you can think about increasing your scores on CAT QA.
Leave no concept unturned
While the QA section of the CAT might seem like one big block of Math, nothing could be farther from the truth.
Each of the topics on CAT QA is a different ballgame altogether and one can’t club it all under a big Math umbrella. This is the reason why test-takers have such varying degrees of expertise across the areas within CAT QA —
– some are exceptional at Numbers but poor at Arithmetic
– some are great at Arithmetic and Geometry but really bad at P&C
– some find P&C and Probability solvable but find functions a problem
This in itself indicates how each topic on Math ends up testing a different kind of mental skill set, making the QA section similar to a Heptathlon or Decathlon, which requires you to be good at 7 and 10 different events.
To compete in such an event you need to first know how to perform in each individual event. You cannot know how to perform only 5 out of 7 events in a heptathlon (100 meters hurdles, High jump, Shot put, 200 meters, Long jump, Javelin throw, 800 meters) and then try to compete.
It goes without saying that to succeed at such an event you need to be above average in all events and great at a few, success on the CAT requires something very similar — you need to know the basics of all the topics and be competent enough to solve Easy and Medium questions from all of them.
So I hope after this no one will ask what the important topics for CAT QA are (that indicates the mindset of Board Exam preparation and not CAT prep).
Once the basics are in place, the three building blocks to get better at CAT QA are: Accuracy, Selection & Speed.
Why accuracy is the first thing you need to work on
The first thing you need to do is to fix the machine or rather ensure that the machine churns out a very high percentage of items within the quality standards. While achieving 6-sigma levels of accuracy is a very high benchmark to set, you should strive to have an accuracy rate of at least 80 percent.
Irrespective of the how many concepts you know, if your machine has an error rate of 35% then you are always going to be performing below par.
- 20 Attempts at 65% accuracy will fetch you 32 marks
- 30 Attempts at 65% accuracy will fetch you 46 marks
- 20 Attempts at 80% accuracy will fetch you 44 marks
- 20 Attempts at 85% accuracy will fetch you 49 marks
What should you focus — attempts or accuracy — given that you are taking the CAT to enter the world of business?
Obviously, accuracy since you will always look to squeeze the maximum out of every dollar invested (unless you run an e-commerce business and have investors to watch your back, albeit not for perpetuity).
What do you think is easier to achieve?
- an increase in attempts from 20 to 30 or
- an increase in accuracy from 65% to 85%
If you accuracy is low then trying to dramatically increase attempts will only further bring down your accuracy. If at your current speed you are prone to crashing 3-4 out of 10 times then at a higher speed you will only crash more often.
So fix the machine to get the most out of it. The table below will give you more than enough reasons to do so.
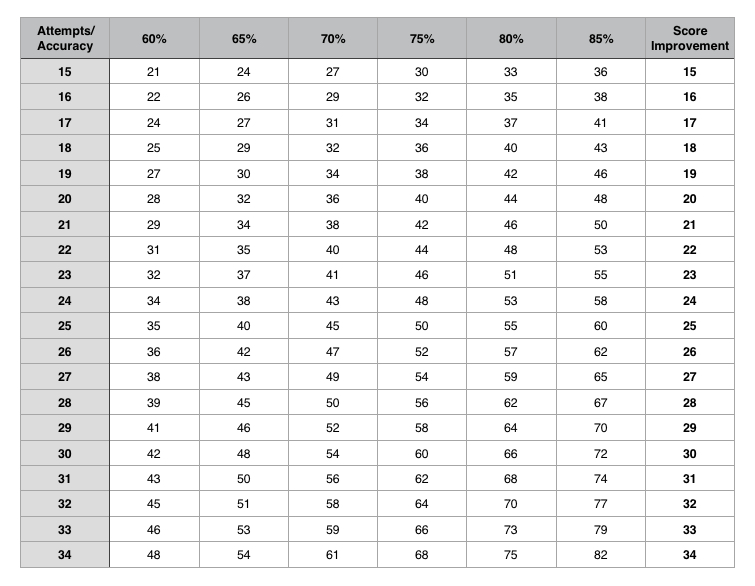
Use the above table to see where you are right now and then try to move rightward first only then try to move downward.
Diagnose the reasons behind your low accuracy
Good accuracy is a function of two things — your solving technique and your choice of questions. Since we will take up selection in the next section of this post, here we will deal with just solving technique.
Since we have undergone the induction process of learning solutions during the long formative years of our education, we don’t really know the technique of problem solving as such. So we usually attribute our mistakes to that worn-out phrase — silly mistake. If we continue to use that phrase then neither can I nor can anyone can else help you out since the only solution is to stop being silly!
Even if you tell yourself that you will be serious, that you will concentrate hard, it is not going to work since there are just words or attitudes and not process changes.
To improve your accuracy on CAT QA, you need to first stop viewing your mistakes through the silly-mistake lens, view it through the process-mistake lens.
These are the big process mistakes to which most errors can be attributed.
Missing crucial information in the question – MISREADING
We are always in a tearing hurry to read the question, so it is not a surprise that we tend to not read the parts of the question, usually the first parts (if n is an integer) or the last part (if they work on alternate days).
Since we are always trying to map a question to a pattern we have previously learned or to a formula, we tend to ignore the unique aspects of the question in front of us and tend selectively pick out information that either matches a pattern or can be put into a formula.
Taking your eye off the ball while calculating – MISCALCULATION
Keen followers of cricket will know how Sunil Gavaskar always gets agitated when a batsman gets run-out because of not grounding the bat. For him it is unpardonable since to ground the bat is part of the process of batsmanship and more importantly, it is a case of throwing away one’s wicket. He is known to have been such a stickler for correctness — he always took an extra run after a century before celebrating since the manual scorer could have made a mistake — no wonder he gets so incensed!
Just like running between the wickets is the hard (or donkey) work in cricket, the calculation part is the hard (or donkey) work in the CAT QA. You can either choose to just run without really being alert and present or be vigilant & fast at the same time a la Dhoni & Virat.
If you watch those two they don’t just run blindly, they have their eye on where the ball has gone and on the fielder, that is what makes them exceptional. They are as alert during the running phase as they are when they are facing up to the ball and playing a shot.
Missing the complexity of the question – MISJUDGEMENT
Sometimes you make a mistake, not because of the above two reasons but because you have underestimated the complexity of a question.
This underestimation can happen at two stages:
- One during the initial stages when you have unknowingly simplified the problem. The reason for this though is again related to mapping a question while reading itself to a previous pattern in your head and thus missing the extra knot that makes the specific question a tad tougher.
- The other during the execution stage in the rush to solve the question and move forward to the next question.
Such errors tend to occur in questions involving permutations & combinations or probability.
So firstly, do a diagnosis of the process mistakes you commit. Make a list of all the mistakes you have made in the preceding SimCATs in an excel sheet and next to each mistake write down the process mistake you made for the questions that you could have solved but ended up messing up.
You will come to know which process mistake is contributing how much to your errors, for example, Misreading (40%), Miscalculation (25%) & Misjudgement (35%)
How to eliminate errors due to misreading
If you are making quite a few errors because of misreading the question and if these questions are not towards the end of a section, which means that the misreading was not due to time constraints then you should:
Drop your pace of reading
It might seem as if you will solve far fewer questions by doing this but dropping the pace does not mean that you should read at a snail’s pace. It just means that you will read without rushing. While you might see a marginal dip in the number of attempts, it will be more than offset by the increase in score.
Read the question in front of you
Do not always map the question in front of you to a pattern or a formula as you read it. This is a big reason why even though you read at the right pace you skip information — you selectively pick and exclude information.
How to eliminate errors due to miscalculation
Different questions will require you to concentrate at different levels, some might take up 20% of your mind space some 80% but the key is that within the solving time of the problem, the same level of concentration has to be maintained, be it 20% or 80%, throughout the solving of the problem without viewing the execution of a solution as burden or taking your eye off the ball during the calculation phase.
Even when you are approximating, which means that you are cutting open something with say three slashes of your sword and not ten, each of the three slashes has to be made with concentration and precision.
Your guard should never drop. You should have a quiet, steady and steely intensity, which is best epitomized by Dhoni and Virat.
How to eliminate errors due to misjudgment
Firstly, these are higher order errors where you are not entirely to blame. The test-setter might have managed to cleverly slip in a trap but that cleverness sometimes relies on you making a process mistake.
So to start off with do not start solving as you start reading. By starting to solve as you read you are setting yourself up for a host of errors:
- taking the wrong thing as X only to calculate it and find it in the answer options, move on to the next question thinking you are right and being shocked when you see the score
- assuming the question to be simple and setting up a simplistic structure to solve and not accounting for the build up in complexity as you are reading the question leading to having to reformulate the problem with different variables and equations
While reading only evaluate how the solution will unfold, what you have what you do not have etc.
Before you jump to the solving just pause for a moment to think about
- the complexity of the problem or possible cases if it is a P&C problem
- what will be convenient — taking X or taking a 100
- what will be convenient taking 100 or taking a number that is a multiple of the ratios (if two things are in the ratio 7:8, and you need to assume the total as some value, it is better to take the total as 15 or 150 and get the two values as 7, 8 or 70, 80 instead taking 100 and getting 700/15 and 800/15)
Improve your solving process
If you see most of our inefficiencies occur because
- we are always in a rush, operating all of the time out of a fear of time running out
- we do not read the question properly, so without figuring out the problem we want to deliver a solution
- we do not think about how to solve the problem, we just jump into solving; aren’t we supposed to think, isn’t this supposed to be a test of reasoning in different contexts?
It is not possible to make these processes changes just like that, you need to program your brain to slip out of its current grooves and create new pathways. To do this talk to yourself before every practice session about the changes you need to make — all the great sportsmen do it.
So before every practice session tell yourself to
- read the question properly till the end without panicking
- concentrate hard and never take your eyes off the ball
- think, think and think and not just regurgitate old solutions.
You cannot do the same thing and expect different results
If the reading of this post has to benefit then you should understand the importance of making these process changes.
When cricketers are a bit out of form and getting out in a particular fashion
- Ricky Ponting getting caught LBW
- Brian Lara getting caught in the slips
- Sachin Tendulkar getting caught driving
What do you think their coaches told them? Did they just say — you are making these silly mistakes just stop making them — they did not?
In two of the cases, they identified a clear process mistake
- Ponting, during that phase, was leaning forward too much and tending to fall over and hence getting trapped in front when the ball was pitching and swinging in
- Lara’s bat was coming down from third slip instead of first slip and given his backlift was resulting in him slashing across the line instead of getting behind it
In other cases, they identified tendencies and just avoided them
- After scoring a paltry 82 runs from 5 innings, including two scores of naught, Sachin came back to score a 241 — batting for more than 10 hours and not playing a single stroke on the off side!
More recently, after a disastrous tour of England, Virat Kohli with the help of Sanjay Bangar identified that the reason for him getting out so frequently to Anderson was that his right toe was pointing towards cover and the left one towards mid-off, making his stance very straight on — this resulted in Anderson squaring him up. So before the first test in Australia, they changed the stance to a more side-on one — right toe pointing towards point and the left towards cover — that helped him play beside the line and leave the ball.
If the best batsmen in the past and current generations can identify process changes and adopt a different approach to get better results, so can you.
Be it in life, in sport, or in business, the mantra is always simple — change or perish!
And for the change to happen do not wait for Newton’s Second Law to kick in; the external force — the need to crack the CAT this year — is already there do not wait for the force to increase — failing to crack it this year — before you make changes.
In the next post we will take up the other two building blocks — selection & speed; before that please do the diagnosis to improve your accuracy and start ringing in the changes.
Pingback: CAT 2018: How to improve your QA percentile – Part II – The CAT Writer
Hello Sir,
I was a classroom student of IMS and gave CAT 2016 and got a score of 93%ile. I am attempting again this year & I have enrolled myself for e-test series of IMS.
I have used the same study material to prepare my basics. After my last SIMCAT score of 60%ile (mainly due to low accuracy) I have taken a break from giving them and trying to solve the Application Builders first. What I notice after your post is that I tend to misjudge the questions and oversimplify them. Is this because I have used the same set of questions to brush my basics again? Do I need to use some other material to start looking at questions with the aim of solving them than asserting them to a particular type?
LikeLike
Hi Sruthi,
If I am not wrong you were a classroom student at IMS Chennai, I think the Annanagar center when you were in college (correct me if I am wrong).
The reason you are misjudging the question is that, as you rightly identified, you are trying to map the problem to a problem you have solved in practice. So the oversimplification is nothing but you ignoring the unique aspect of the question in front of you.
While basics are the same you need to always every question, which will primarily be an application question, as you would look at a brand new question and not look to replicate a solution. This does not mean that the practice won’t help but just that the question will always have a slight variation else everyone with a good memory will crack the test.
Hope this clarifies,
All the best!
LikeLike
Thank you for your reply, Sir. I have altered my approach according to your suggestion and I have seen a slight improvement in accuracy.
I was actually a student of IMS Powai in Mumbai because I was working there after graduation. I have shifted it to Chennai now.
LikeLike
Thank you for your reply, Sir. I have altered my approach according to your suggestion and I have seen a slight improvement in accuracy.
I was actually a student of IMS Powai in Mumbai because I was working there after graduation. I have shifted it to Chennai now.
LikeLike